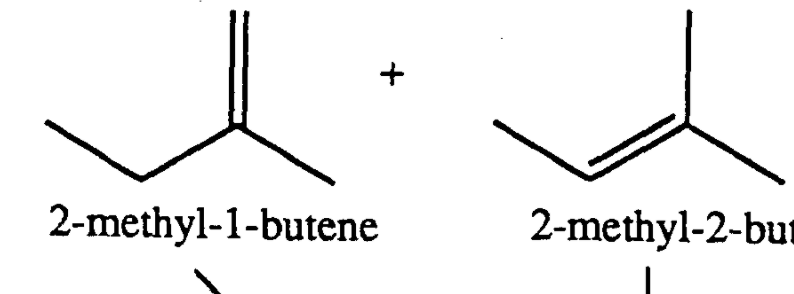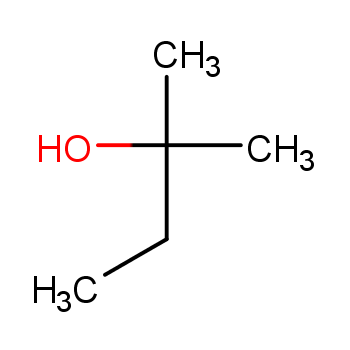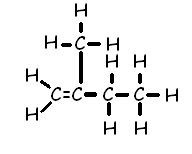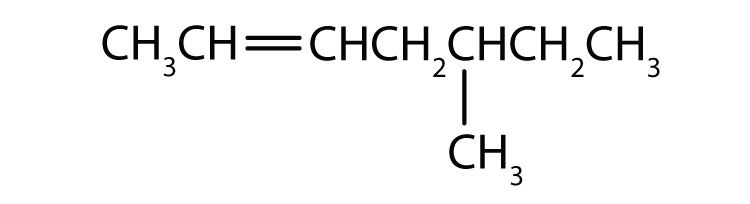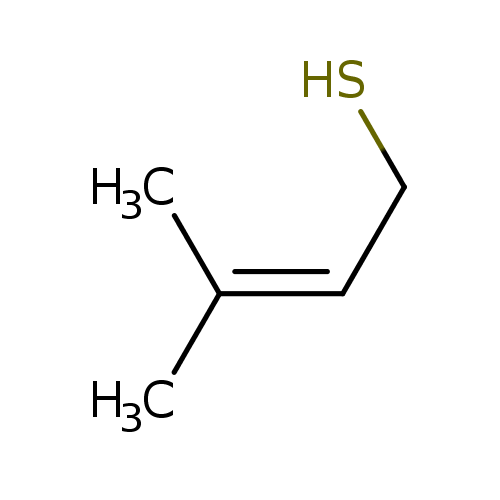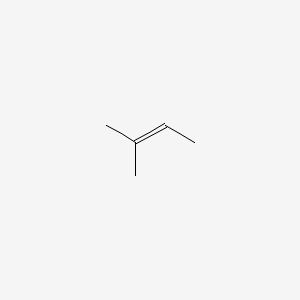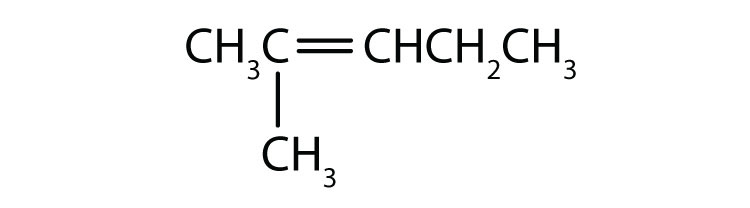
Chapter 1 - Organic Chemistry Review / Hydrocarbons - CHE 120 - Introduction to Organic Chemistry - Textbook - LibGuides at Hostos Community College Library

Give the condensed structural formula for each of the following compounds. a. 3-ethyl-2-pentene. b. 4-ethyl-2-methyl-2-hexene. | Homework.Study.com

Predict the major and minor products of 2-methyl-2-butene with HBr as an electrophilic addition reaction. Include the intermediate reactions. | Homework.Study.com
![The compound ${{2 - methyl - 2 - butene}}$ on reaction with \\[NaI{O_4}\\] in the presence of \\[KMn{O_4}\\] gives:\n \n \n \n \n \tA.\\[C{H_3}COOH\\]B.\\[C{H_3}COC{H_3} + C{H_3}COOH\\]C.\\[C{H_3}COC{H_3} + C{H_3}CHO\\]D.\\[C{H_3}CHO + C{O_2}\\] The compound ${{2 - methyl - 2 - butene}}$ on reaction with \\[NaI{O_4}\\] in the presence of \\[KMn{O_4}\\] gives:\n \n \n \n \n \tA.\\[C{H_3}COOH\\]B.\\[C{H_3}COC{H_3} + C{H_3}COOH\\]C.\\[C{H_3}COC{H_3} + C{H_3}CHO\\]D.\\[C{H_3}CHO + C{O_2}\\]](https://www.vedantu.com/question-sets/36531b4a-2453-4b90-9e48-61c2438889662404814985533969518.png)